
The following is included in this e-tech newsletter:
- Bioplastics in drug delivery dosage forms - Making lives better!
- 2 extruders in 1: ZSE-16 process section attachment
- Twin screw granulation (TSG) helps transition from batch to continuous processing
- Counter-rotating intermeshing twin screw technologies revisited for life science applications
Bioplastics in drug delivery dosage forms- Making lives better!
Plastics plays a role in all facets of modern life, with billions and billions of pounds utilized to make consumer goods that we use every day. Bioplastics, derived from renewable resources, represent an evolution within plastics that are making a big difference in the quality of countless people's lives! High-performance plastics are also used for a variety of nutritional, medical and pharmaceutical applications, including FDA approved water soluble polymers that facilitate controlled release drug of delivery products to humans and animals (i.e. tablets, capsules, transdermal patches, etc.). Bioplastics are beginning to play a prominent role in this space, particularly for implantable and multi-functional devices such as sutures, insertable wafers, ocular delivery fibers, 3D filaments and other non-traditional drug delivery systems.

ZSE-3D filament system for prototype drug delivery devices
The degradable nature of bioplastics is ideal for implantation into the body for localized treatment and relief. Non-degradable delivery implants (i.e. silicon implantable contraceptive devices) generally require surgical removal at the termination of the dosing regimen. It's obvious a 2nd procedure for any implanted device is undesirable for several reasons: cost, procedural complications, risk of infection and the possibility of lack of compliance. The use of bioplastics that degrade under physiologic conditions presents many advantages. For instance, PLA and PLGA can act as drug depot polymers that are mixed with drugs and additives via twin screw extrusion (without over-shearing and degrading the drug or polymer) to impart the desired drug release and erosion properties from the implanted device.
The degradable nature of bioplastics is ideal for implantation into the body for localized treatment and relief. Non-degradable delivery implants (i.e. silicon implantable contraceptive devices) generally require surgical removal at the termination of the dosing regimen.
It's obvious a 2nd procedure for any implanted device is undesirable for several reasons: cost, procedural complications, risk of infection and the possibility of lack of compliance. The use of bioplastics that degrade under physiologic conditions presents many advantages. For instance, PLA and PLGA can act as drug depot polymers that are mixed with drugs and additives via twin screw extrusion (without over-shearing and degrading the drug or polymer) to impart the desired drug release and erosion properties from the implanted device.

The use of twin screw extruders offers significant advantages as compared batch and continuous mixers. After 100 or so years of usage in the plastics industry, the well-characterized nature of the twin screw extrusion process lends itself to process optimization, while also affording benefits of continuous manufacturing, which is preferred in the current regulatory and fiscal environment (ref. FDA Process Analytical Technology Initiative, 2004). There are many possibilities to incorporate drugs into biodegradable polymers for unique dosage forms, including but not limited to dissolvable films, ocular delivery devices, and foamed extrudates. The success of numerous commercial products has prompted the scientific community to expand and explore the use of bioplastics as novel dosage forms.
Development efforts continue to expand for non-traditional drug delivery mechanisms. These products represent a small percentage of the bioplastics being used today, but the positive impact to society is significant. And there's more to come!
2 extruders in 1: ZSE-16 process section attachment
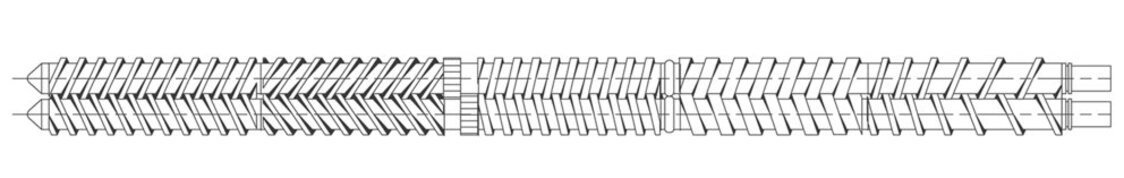
The Leistritz ZSE 16 twin screw extruder's process section is designed to process 20 to 100 gram batches and can be supplied as a stand-alone extrusion system or mated to a new or existing ZSE-18 gearbox. Like any twin screw extruder, screws and barrels are modular and designed for efficient mixing. Low free volume (inherent with a tri-lobal design) combined with ultra-high torque (due to a splined shaft design) facilitate sampling and feasibility testing not possible on other twin screw extrusion devices.
Tri-lobal design - end view

ZSE-16 twin screw extruder system
The ZSE-16 process section attaches to new or existing ZSE-18 gearbox to facilitate ultra-low rate processing and batch sampling of 100 grams or less. The following specifications apply:
- 16 mm screws dia., 1 mm flight depth, 1.2/1 OD/ID ratio
- 72 NM torque, 1 cc/rev. free volume
- Segmented ss screws on splined shafts
- Segmented 2-piece ss barrel design · Water cooled feed barrel
- 2-zone 16/1 L/D ss barrel w/ internal cooling bores
- Specialty feed inserts ultra-low rate feeding
- HMI screen for ZSE-16 processing
Please contact us today for a quotation for a stand-alone ZSE-16 or to retrofit a ZSE-16 onto an existing ZSE-18 twin screw extruder.
Twin screw granulation (TSG) helps transition from batch to continuous processing
The use of twin screw granulation (TSG) as part of the manufacturing process for tablets and capsules is increasing, as is reflected by the number of process patents and products utilizing the TSG manufacturing platform. In addition to the pharmaceutical and nutrient markets, TGS is also used in other industries to prevent material segregation and to improve product flowability, compressibility and myriad of other properties.

High-shear batch granulators consist of mixing blades positioned inside large mixing chambers. Batch granulation operates under globally starved and unpressurized conditions. Powder is randomly subjected to the high shear zone in a batch granulator, with the potential for non-uniform mixing dynamics. In the TSG continuous process the material is conveyed through a sequence of starved (partially filled flights) and pressurized (non-pumping mixers) zones dictated by the screw element selection. The incoming formulation is essentially in a first-in, first-out process with a consistent and repeatable residence time and residence time distribution, which ensures the consistent quality of granules and a uniform mixing history.
The twin screw extruder is an extremely versatile tool that can be used for TSG (melt or wet) as well as melt extrusion. Advantages of TSG: (1) more homogeneous distribution of drug/excipient/ binder; (2) reduced level of binder to achieve the desired granules; and (3) significantly shorter processing time (10-20 seconds or less). A short residence times makes it feasible to explore high processing temperatures than is practical for a batch process. Additional benefits include: smaller footprint, less capital investment, and real-time monitoring (rather than postproduction testing).

Click on the cover for the article. This article highlights: granule formation mechanisms, benefit over conventional batch processes, process control and monitoring, TSG history, and anticipated TSG developments.
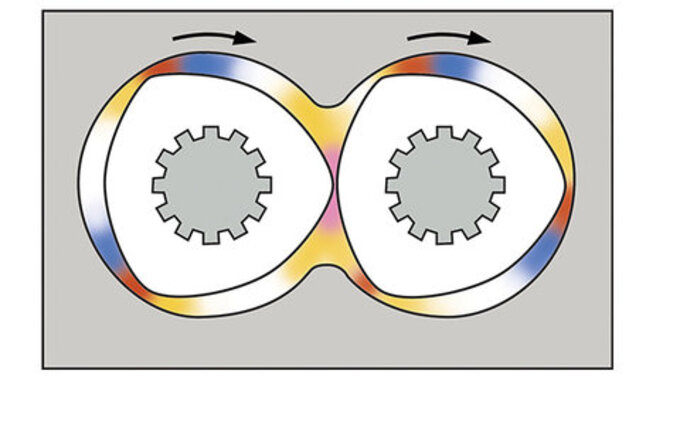
Counter-rotating intermeshing twin screw technologies revisited for life science applications
Counter-rotating twin screw extruders, where the screws rotate in opposite direction, have been a viable extrusion technology since the 1930s. There are 2 different types of counter-rotating twin screw extruders:
Low speed, late fusion (LSLF) intermeshing mode is designed to avoid energy input and do not melt the materials until the middle or latter part of the process section. The LSLF counter-rotating twin screw extruder is characterized by a gentle melting and mixing effect and narrow residence time distribution in combination with high pressure pumping capabilities. Comparatively low screw rpms (less than 50) and late fusion screws are designed to avoid imparting too much energy into the process.
Counter-rotating, intermeshing screw set
High speed, energy input (HSEI) intermeshing mode is designed as a mass transfer device and shares many of the characteristics of the more widely utilized co-rotating intermeshing mode of operation, including modular process section with efficient heating and cooling design. Primary applications include compounding, specialty reactions and devolatilization.

Close-up hexa-lobal, counter-rotating elements
In either mode, only counter-rotating twin screw extruders can be designed to pump materials in a non-drag flow manner in locked C-shaped chambers, as compared to the drag flow single screw extruder and semi-drag flow co-rotating twin screw extruder. This allows tight tolerance parts to be produced directly from the counter-rotating intermeshing extruder without a gear pump.
A number of interesting studies have been performed in counter-rotation, see these links for more details:
- General Characteristics of Counter-rotating Twin Screw Extruders
- Investigating the Impact of Extruder Process Parameters and Rotation Mode on HPMCAS Degradation
- Comparison of Various Operating Conditions and Formulations on a Counter-rotating, Intermeshing Twin Screw Extruder
- Investigation of Process Temperature and Screw Speed on Properties of a Pharmaceutical Solid Dispersion using Co-rotating and Counter-rotating Twin-screw Extruders
Counter-rotating, intermeshing twin screw technologies are available for testing in the Leistritz NJ process laboratory.
For additional information on anything contained in this newsletter call 908/685-2333 or e-mail sales@leistritz-extrusion.com
Your Leistritz team,
Leistritz Extrusion
175 Meister Ave. Somerville, NJ, 08876, USA
ph: 908/685-2333
email: sales@leistritz-extrusion.com