
The following is included in this e-tech newsletter:
- Video link: Investigating the Impact of Twin Screw Extruder Process Parameters and Rotation Mode on HPMCAS Degradation
- Leistritz Pharmaceutical-Nutraceutical Extrusion Seminar: June 12-13, 2019 in NJ
- Featured article: Processes, challenges and the future of twin screw granulation for manufacturing oral tablets and capsules
- PharmSci360 to be held in San Antonio, TX
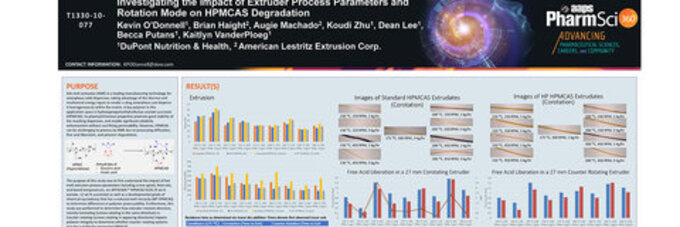
Check Investigating the Impact of Twin Screw Extruder Process Parameters and Rotation Mode on HPMCAS Degradation
Melt extrusion (ME) is a leading manufacturing technology for amorphous solid dispersions, taking advantage of the thermal and mechanical energy inputs inherent with a twin screw extruder to render a drug amorphous and disperse it homogeneously within the polymer matrix. A key polymer in this application space is hydroxypropylmethylcelluloseacetate succinate (HPMCAS). Its physical/chemical properties promote good stability of the resulting dispersion, and enable solubility enhancement without sacrificing permeability. However, HPMCAS can be challenging to process via extrusion due to processing difficulties, free acid liberation, and polymer degradation.
The purpose of this study was to first understand the impact of melt extrusion process parameters including screw speed, feed rate, and barrel temperatures on AFFINISOL™ HPMCAS 912G as well as a developmental grade of chemical equivalency that has a reduced melt viscosity (HP HPMCAS) to determine differences in polymer processability. Furthermore, this study was performed to determine how extruder rotation direction, namely co-rotating (screws rotating in the same direction) vs. counter-rotating (screws rotating in opposing directions) impacts polymer integrity.
Check out a video summary by Kevin O'Donnell, R&D manager, DuPont Nutrition and Health.
HERE you can download a free pdf of the study.
Leistritz Phamaceutical-Nutraceutical Seminar
June 12-13, 2019
Special emphasis on melt extrusion, multi-functional devices and nutritional products
Leistritz will host the annual Pharmaceutical-Nutraceutical Extrusion Seminar on June 12-13, 2019. This "hands-on" seminar starts with the basics and explains why twin screw extrusion is a preferred manufacturing method to manufacture consistent, repeatable products.
In addition to Leistritz staff, outside experts from industry and academia will contribute to the program.
Everything you need to know you will find HERE.
Processes, challenges and the future of twin screw granulation for manufacturing oral tablets and capsules
Adopting twin screw granulation can aid in the transition from batch to continuous processes. The majority of tablets and capsules products are manufactured using granulation processes including wet, melt, and dry granulation. These granulation techniques are also commonly used across many other industries, including food and nutrient industries. Powders are granulated to prevent material segregation and to improve powder properties, including flowability and compressibility. While other industries transitioned wet and melt granulation from batch to continuous processes in the 1970s, the adoption of twin-screw granulation (TSG) has been slow in the pharmaceutical industry.
As pharmaceutical scientists became more familiar with TSG, an increasing number of process patents and products utilizing the TSG platform is anticipated. This article highlights four aspects of TSG: (a) granule formation mechanisms, (b) benefit over conventional batch processes, (c) process control and monitoring, and (d) history, recent progresses, and the future.
For a pdf of the entire article as it appeared in AAPS March 2018 news magazine please CLICK HERE.

ZSE-27 twin screw extruder configured for granulation
Don't miss this event!
Visit Leistritz at Pharm360 in San Antonio, TX on November 3-6, 2019!
Your Leistritz team
Leistritz Extrusion
175 Meister Ave.
Somerville, NJ, 08876, USA
ph: 908/685-2333
email: sales@leistritz-extrusion.com
www.leistritz-extrusion.com